|
A standard 1 GW nuclear fission plant requires 150,000 Mt uranium-containing ore to fabricate enough fuel for 1 year. The isotope uranium-238 accounts for over 99% of the uranium in nature, but nuclear fission fuel requires the uranium-235 isotope. Uranium rests in geologic strata in the minerals uraninite and pitchblende. Nuclear fission plants require highly processed uranium fuel. The similarity of the atomic structure of strontium to calcium increases the uptake of strontium by animals and its incorporation into bones.  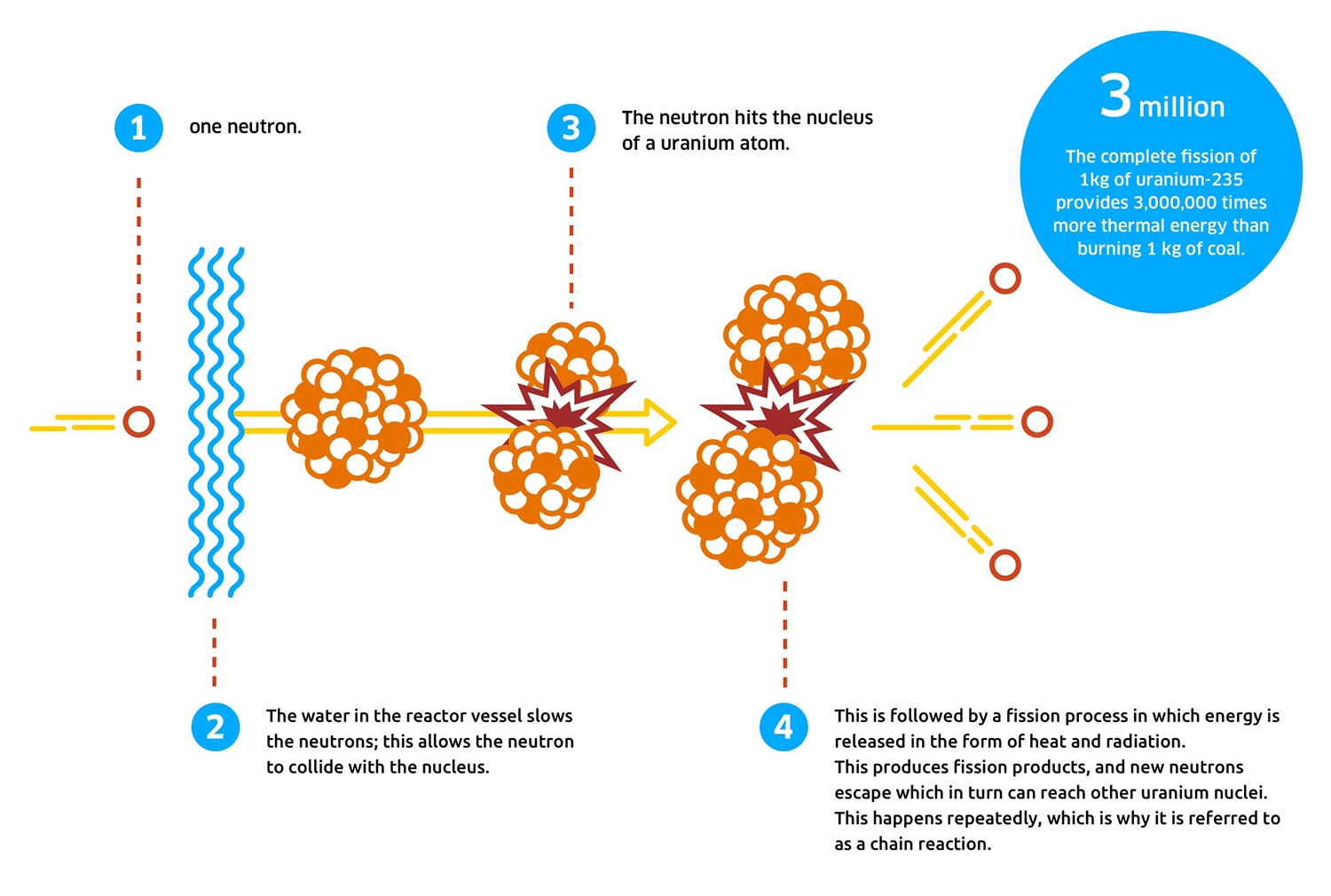
While the half-life of strontium-90 is 29 years and the half-life of cesium-137 is 30 years, plutonium-239 decays with a half-life of 25,000 years, and a quantity of iodine-129 will decay to half of its mass only after 17 million years. When radiation passes through living tissue, the particles or photons impart their energy to atoms and molecules in the tissue, disrupting molecular and atomic structures.įission products emit radiation until they reach a stable atomic state. Radiation consists of kinetic energy of small molecules and atomic particles and electromagnetic energy of photons traveling at certain frequencies. A fission reaction produces energy in the form of light, heat, motion of the fission products, and radiation. 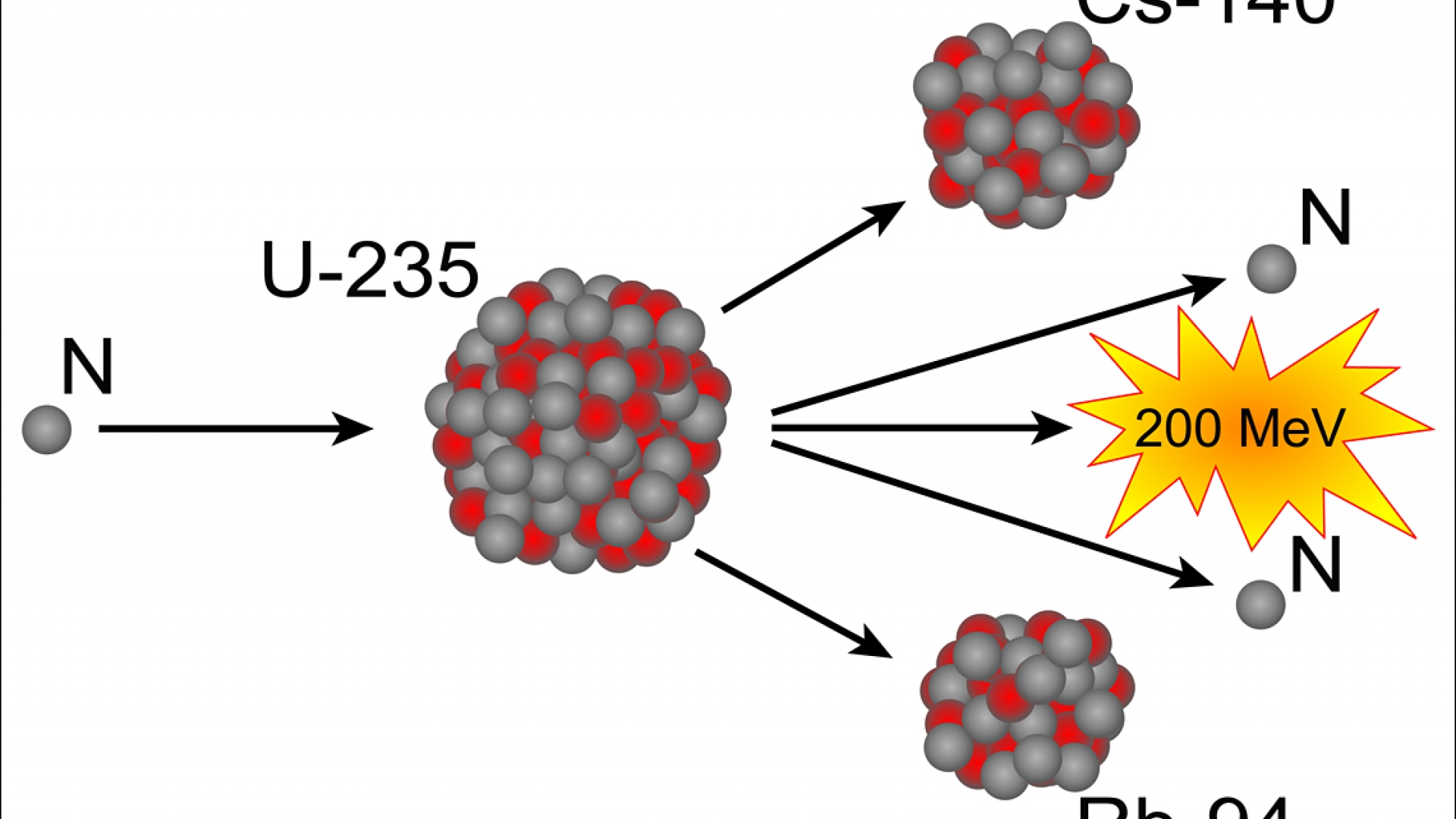
Uranium and plutonium are the elements that provide the most effective yield from fission. Nuclear fission is the splitting of high-molecular-weight elements to release energy held by protons and neutrons in the nucleus of the atom. Patrick Gonzalez, in Encyclopedia of Biodiversity (Second Edition), 2013 Nuclear Fission
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
